Evaluation of Sulfide Removing Rate by Basic Zinc Carbonate in Oil Based Drilling Fluids and Analyses of Sulfide Removing Mechanisms
-
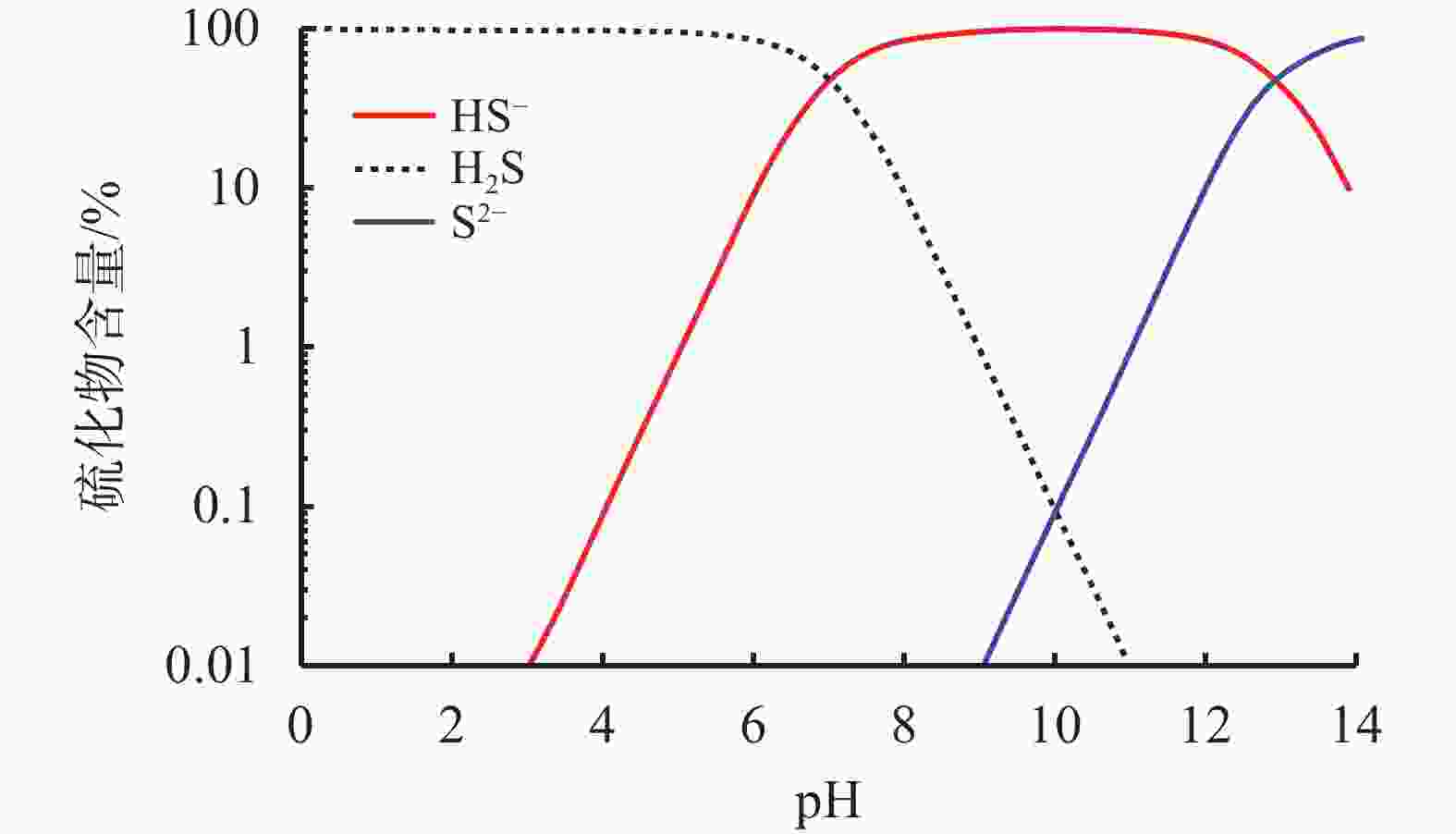
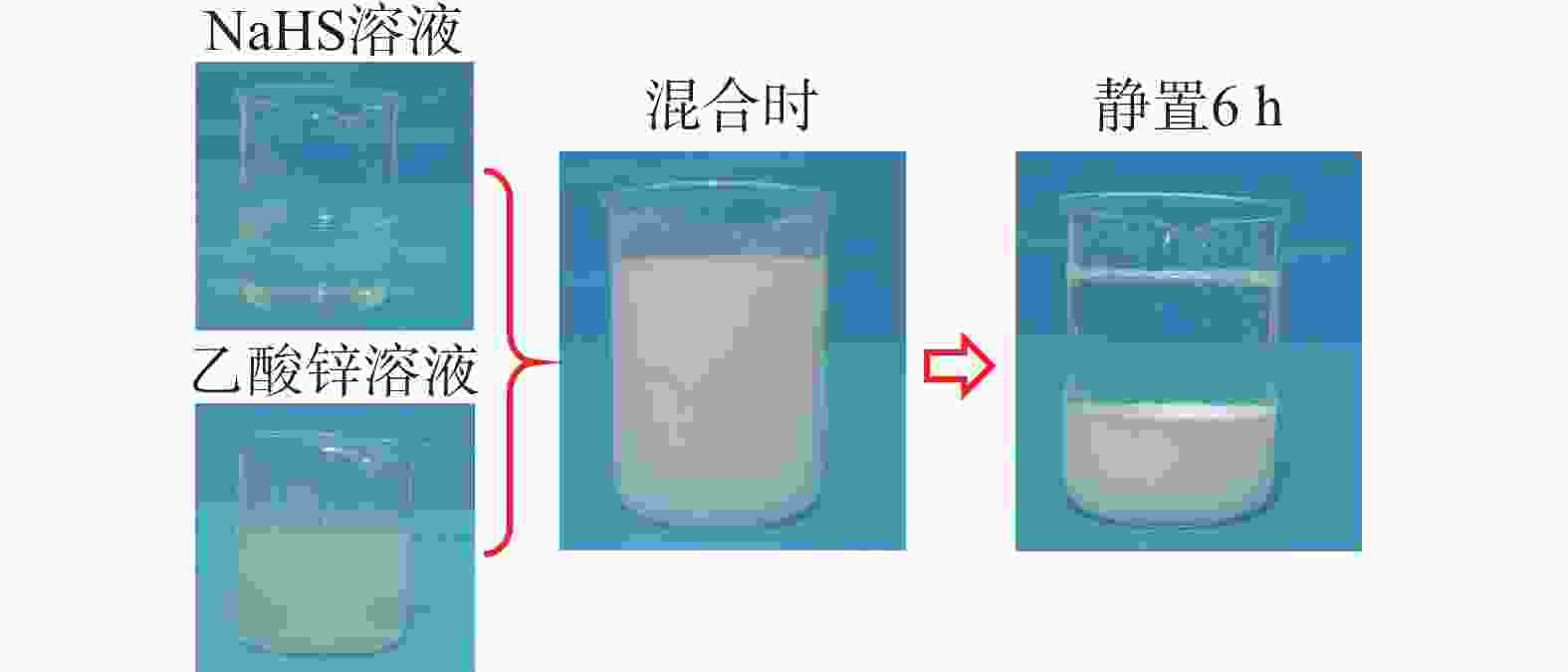
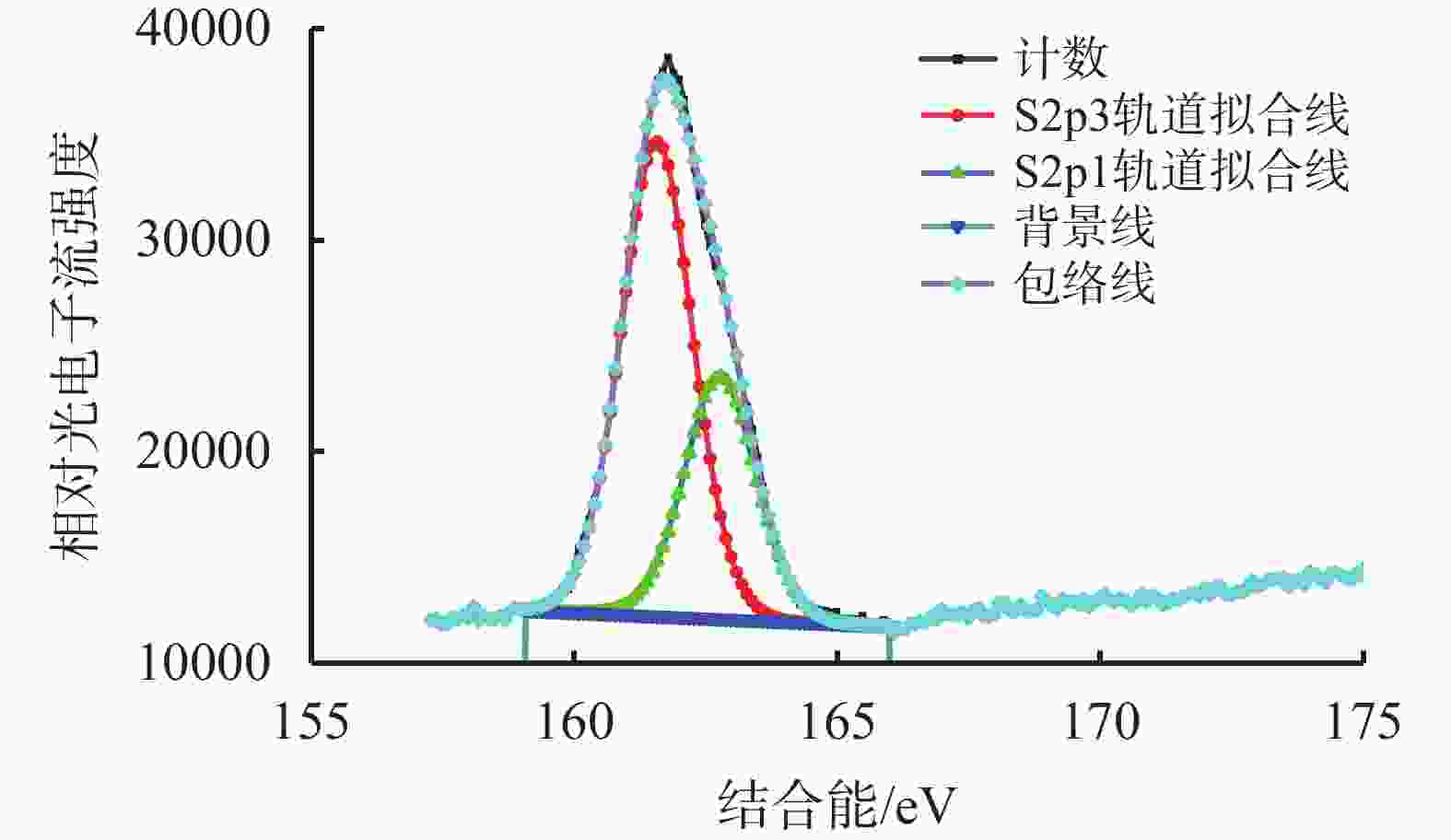
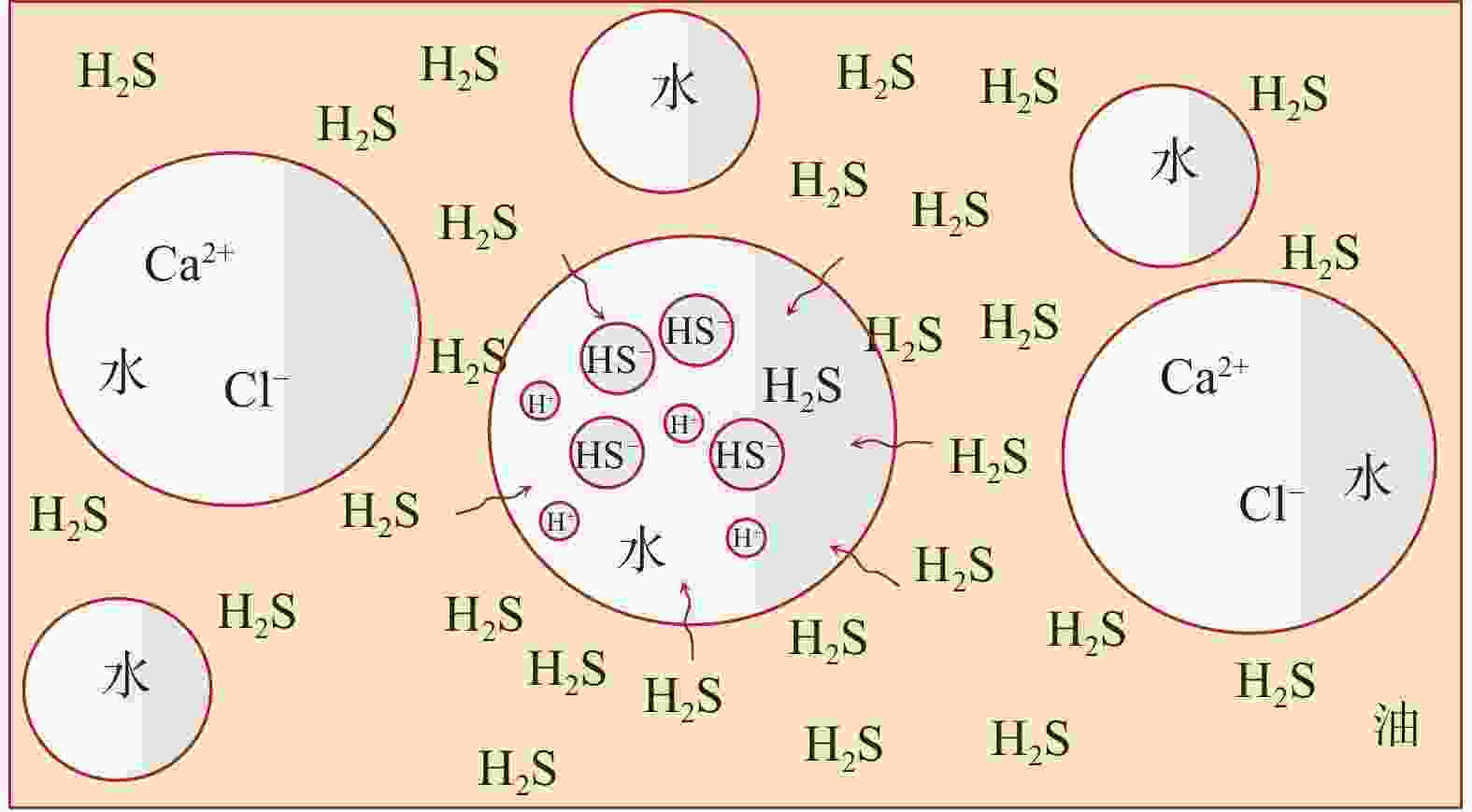
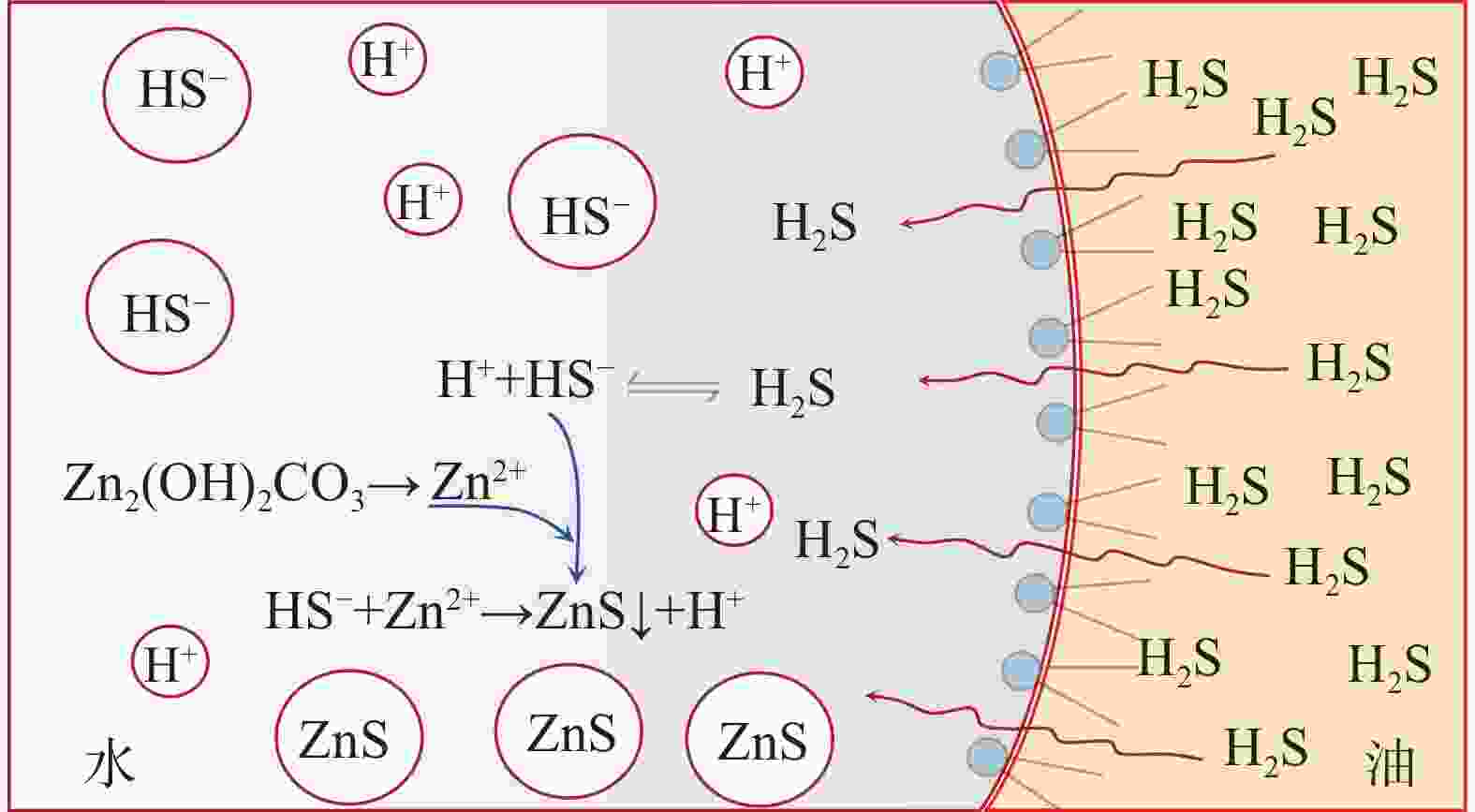
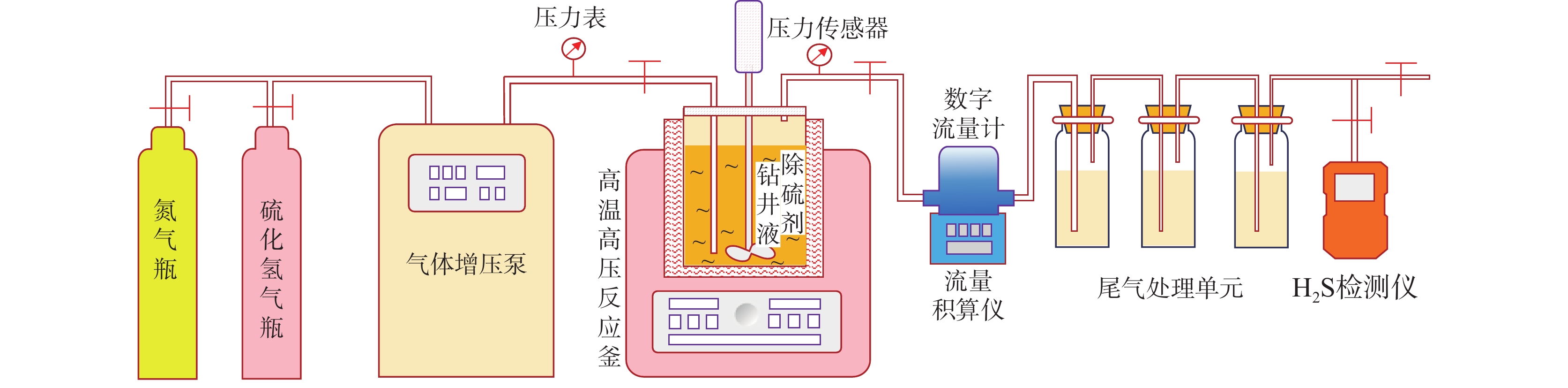
摘要: 碱式碳酸锌是高含硫地层水基钻井液常用除硫剂,其在油基钻井液中的除硫能力和作用机制尚不明晰。针对传统评价方法中硫化氢浓度和流量偏低、适用温度不高、反应时间过短等不足,搭建了适用于深井钻井液除硫率评价的实验平台;利用钻井液除硫率评价实验平台,测试了碱式碳酸锌在油相、水相、油包水乳液及油基钻井液中的除硫率;基于除硫率实验结果,分析了硫化氢在油包水乳液中的存在形式,厘清了中性及弱碱性水相环境中碱式碳酸锌除硫反应机制,进而揭示了油基钻井液中碱式碳酸锌除硫作用机制。结果表明,油基钻井液中碱式碳酸锌除硫率可达100%;侵入油基钻井液的硫化氢大部分(>90%)以未离解硫化氢分子形式存在于油相中,少部分硫化氢(<10%)溶解于水相中并离解形成以HS−为主的离子;水相中碱式碳酸锌电离出的Zn2+离子直接与硫化氢一级电离产物HS−离子发生反应生成ZnS沉淀,水相高pH值并非为除硫的必要条件;反应-扩散耦合作用是油基钻井液中碱式碳酸锌的主要机制;温度升高有助于提高除硫效率。揭示的油基钻井液中碱式碳酸锌除硫作用机制,为碱式碳酸锌用作油基钻井液除硫剂提供了科学依据。Abstract: Basic zinc carbonate is a commonly used sulfide scavenger for drilling high-sulfide content formations with water-based drilling fluids, its ability to remove sulfide and the mechanism of sulfide removal in oil-based drilling fluids are still not well understood. In evaluating the rate if hydrogen sulfide removal, conventional methods use relatively low concentration and flow rate of hydrogen sulfide, the results of these methods are not suitable for high temperature application, and the reaction time in these methods is too short. To overcome these deficiencies of the old evaluation methods, a new experimental platform for evaluating the rate of sulfide scavenging of deep-well drilling fluids has been constructed. Using this platform, the rates of sulfide scavenging of basic zinc carbonate in oil, water, water-in-oil emulsion and oil-based drilling fluid were tested; the test results were used to analyze the existence forms of hydrogen sulfide in water-in-oil emulsions, the sulfide scavenging mechanisms of basic zinc carbonate in neutral and weakly-alkaline aqueous phases were clarified, and the sulfide scavenging mechanism of basic zinc carbonate in oil-based drilling fluids was then revealed. The test results show that the rate of sulfide scavenging of basic zinc carbonate in oil-based drilling fluids can be as high as 100%; most of the hydrogen sulfide (>90%) invading into an oil-based drilling fluid exists as undissociated hydrogen sulfide molecules in the oil phase of the oil-based drilling fluid, and only a small fraction (<10%) of the invading hydrogen sulfide dissolves into the aqueous phase to form ions dominated by HS−. In the aqueous phase of an oil-based drilling fluid, the Zn2+ ions ionized from basic zinc carbonate react directly with the primary ionization product HS− of hydrogen sulfide to form ZnS precipitate, and a high pH of the aqueous phase is not a necessary condition for sulfide scavenging; the reaction-diffusion coupling effect is the primary mechanism of sulfide scavenging by basic zinc carbonate in oil-based drilling fluids, and temperature increase is helpful to enhance the rate of sulfide scavenging. The revealed working mechanism of basic zinc carbonate in oil-based drilling fluids provides a scientific basis for using this chemical as a sulfide scavenger in oil-based drilling fluids.
-
表 1 不同液体中碱式碳酸锌除硫率实验
液体 Vs/
mLVp/
mLVh/
mLVr/
mLη/
%水 1636 480 464 1056 64.56 水+3%碱式
碳酸锌1725 370 0 1630 94.51 白油 1829 281 401 1318 72.05 白油+3%碱式
碳酸锌1754 312 340 1348 76.84 油包水乳液 1771 1084 338 828 46.75 油包水乳液+
3%碱式碳酸锌1771 684 0 1566 88.42 油基钻井液 1292 1404 45 801 61.99 油基钻井液+
3%碱式碳酸锌1484 981 0 1269 85.53 表 2 温度对碱式碳酸锌除硫率的影响
样品 T/℃ p/MPa η/% 油基钻井液 25 0.10 61.99 油基钻井液+
3%碱式碳酸锌25 0.10 85.53 120 0.27 93.96 150 0.34 96.14 180 0.72 96.53 表 3 不同流体中硫化氢溶解度实验结果
液体 Vs/
mLVp/
mLVr/
mL常温常压溶解度/
mL/mL油 1829.2 110.2 1719 2.87 水 1635.6 115.6 1520 2.53 25%CaCl2水溶液 1252.9 551.9 701 1.17 W/O乳液(90∶10) 1806.4 156.4 1650 2.75 -
[1] 耿东士, 何纶, 李道芬, 等. 钻井液中硫化氢的危害及其控制[J]. 钻井液与完井液, 2007, 24(增刊1): 1-3.GENG Dongshi, HE Guan, LI Daofen, et al. Hazards of hydrogen sulfide in drilling fluids, and the control thereof[J]. Drilling Fluid & Completion Fluid, 2007, 24(S1): 1-3. [2] 卫伟, 张洁, 朱宝忠, 等. 钻井液用除硫方法研究进展[J]. 化工技术与开发, 2020, 49(1): 25-30, 41.WEI Wei, ZHANG Jie, ZHU Baozhong, et al. Research progress of desulfurization method for drilling fluid[J]. Technology & Development of Chemical Industry, 2020, 49(1): 25-30,41. [3] 周杨, 杨桢剑, 黄宁生, 等. 钻井液中硫化氢污染特点及高效除硫剂性能评价[J]. 石油化工应用, 2016, 35(4): 33-36, 46.ZHOU Yang, YANG Zhenjian, HUANG Ningsheng, et al. The drilling fluid pollution characteristics of hydrogen sulfide and performance evaluation of high efficiency sulfur removal agent[J]. Petrochemical Industry Application, 2016, 35(4): 33-36,46. [4] 陈传濂, 黄纹琴. 钻井泥浆除硫剂的研究[J]. 石油学报, 1982(4): 49-58.CHEN Chuanlian, HUANG Wenqin. Researches on sulfide scavenger for drilling fluids[J]. Acta Petrolei Sinica, 1982(4): 49-58. [5] 肖金裕, 周华安, 暴丹, 等. 川东北飞仙关高含硫地层油基钻井液复合除硫技术[J]. 钻井液与完井液, 2024, 41(3): 350-356.XIAO Jinyu, ZHOU Huaan, BAO Dan, et al. Use of compound desulfurizing agent in high sulfur Feixianguan formation drilling in northeast Sichuan with oil based drilling fluids[J]. Drilling Fluid & Completion Fluid, 2024, 41(3): 350-356. [6] 肖金裕, 周华安, 暴丹, 等. 川东北高含硫气藏钻井液抗硫工艺优化与应用[J]. 钻井液与完井液, 2023, 40(6): 718-724.XIAO Jinyu, ZHOU Huaan, BAO Dan, et al. Optimization of sulfur-resistant drilling fluid techniques and its application in drilling high sulfur content reservoirs in northeast Sichuan[J]. Drilling Fluid & Completion Fluid, 2023, 40(6): 718-724. [7] 贺立勤, 王军, 简旭, 等. 铁山坡高含硫气藏安全快速钻井技术优化与应用[J]. 钻采工艺, 2024, 47(3): 49-57.HE Liqin, WANG Jun, JIAN Xu, et al. Optimization and application of safe and fast drilling technology in Tieshanpo gas reservoir with high sulfur[J]. Drilling & Production Technology, 2024, 47(3): 49-57. [8] 暴丹, 周华安, 刘思源, 等. 油基钻井液高效除硫体系优化实验[J]. 石油与天然气化工, 2023, 52(4): 66-72.BAO Dan, ZHOU Huaan, LIU Siyuan, et al. Optimization experiment of efficient hydrogen sulfide removal system for oil-based drilling fluid[J]. Chemical Engineering of Oil and Gas, 2023, 52(4): 66-72. [9] 李树刚, 魏振吉, 孙中磊. 钻井液除硫剂除硫效果的静态评价实验及认识[J]. 天然气工业, 2012, 32(8): 82-87.LI Shugang, WEI Zhenji, SUN Zhonglei. Static evaluation of indoor experimental conditions for the effect of desulphurization agents in drilling fluids[J]. Natural Gas Industry, 2012, 32(8): 82-87. [10] 刘榆, 李道芬, 孔传明, 等. 钻井液用除硫剂的除硫效率测定方法研讨[J]. 钻井液与完井液, 2007, 24(增刊1): 4-5.LIU Yu, LI Daofen, KONG Chuanming, et al. A discussion on the measurement of cop of the sulfide scavengers[J]. Drilling Fluid & Completion Fluid, 2007, 24(S1): 4-5. [11] 王宝辉, 张丽华, 陈新萍, 等. 石油钻井过程硫化氢污染和化学控制技术的研究与发展[J]. 中国安全科学学报, 1998, 8(3): 6-9.WANG Baohui, ZHANG Lihua, CHEN Xinping, et al. Study and advance on H2S pollution and its control technology in the process of oil well drilling[J]. China Safety Science Journal, 1998, 8(3): 6-9. [12] 万立夫, 李根生, 田守嶒, 等. 钻井过程中硫化氢侵入井筒后的赋存状态研究[J]. 石油钻探技术, 2013, 41(6): 29-33.WAN Lifu, LI Gensheng, TIAN Shouceng, et al. Occurrence state of hydrogen sulfide invading into wellbore during drilling[J]. Petroleum Drilling Techniques, 2013, 41(6): 29-33. [13] BURGER D E, JENNEMAN G D, CARROLL J J, et al. On the partitioning of hydrogen sulfide in oilfield systems[C]//Paper presented at the SPE International Symposium on Oilfield Chemistry. The Woodlands, Texas, USA: SPE, 2013: SPE-164067-MS. [14] ZEA L, COOPER D, KUMAR R, et al. Hydrogen sulfide absorption phenomena in brine/oil mixtures[J]. SPE Journal, 2011, 16(4): 931-939. doi: 10.2118/145401-PA [15] RINKER B E, SANDALL O C, et al. Physical solubility of hydrogen sulfide in several aqueous solvents[J]. The Canadian Journal of Chemical Engineering, 2000, 78(1): 232-236. doi: 10.1002/cjce.5450780130 [16] FAUVE, R, GUICHET X, LACHET V, et al. Prediction of H2S solubility in aqueous NaCl solutions by molecular simulation[J]. Journal of Petroleum Science and Engineering, 2017, 157: 94-106. doi: 10.1016/j.petrol.2017.07.003 [17] CUEVASANTA, E, DENICOLA A, ALVAREZ B, et al. Solubility and permeation of hydrogen sulfide in lipid membranes[J]. PLoS One, 2012, 7(4): e34562. doi: 10.1371/journal.pone.0034562 [18] NIETO DRAGHI C, Mackie A D, BONET AVALOS J, et al. Transport coefficients and dynamic properties of hydrogen sulfide from molecular simulation[J]. The Journal of chemical physics, 2005, 123(1): 014505. doi: 10.1063/1.1949208 [19] ROMERO I, MONTERO F, KUCHERYAVSKIY S, et al. Temperature-and pH-dependent kinetics of the aqueous phase hydrogen sulfide scavenging reactions with MEA-triazine[J]. Industrial & Engineering Chemistry Research, 2023, 62(21): 8269-8280. -



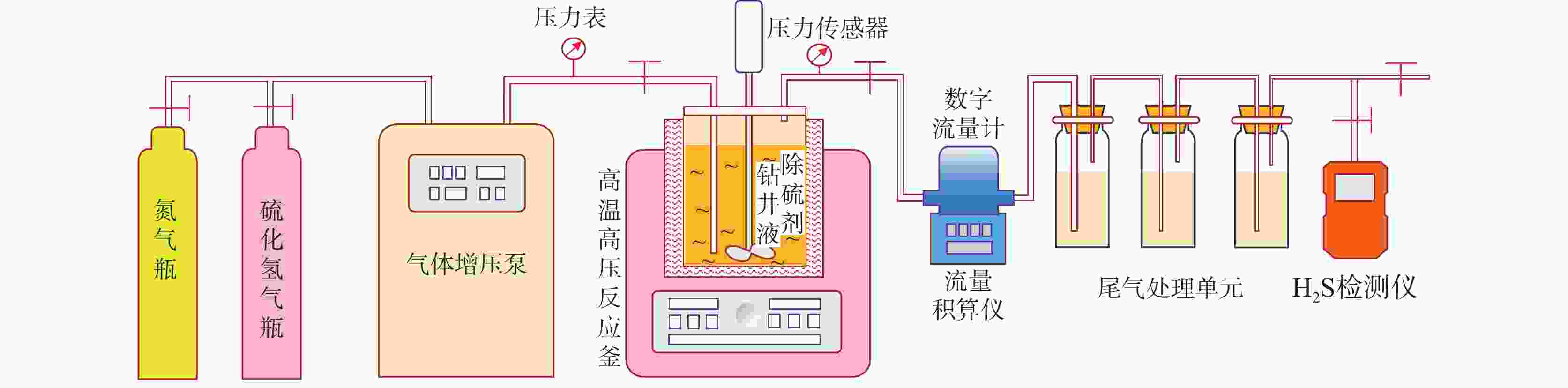
 下载:
下载: